The Clinical Trials — What is Known About the Long-Term Effects of Rexulti?
 Although it is classified as an antipsychotic drug, one of the common uses of Rexulti is to prescribe it on top of antidepressant medications when the antidepressants that were being used failed to get a positive change in the person. The majority of drug trials on Rexulti have been to study efficacy in treating major depressive disorder. The FDA granted approval for this use when Rexulti first appeared on the market. Notably, much research shows that only a percentage of persons with MDD adequately respond to antidepressant medications, according to a mammoth review of medical literature published in 2015 in the Journal of Psychiatry and Neuroscience.1
Although it is classified as an antipsychotic drug, one of the common uses of Rexulti is to prescribe it on top of antidepressant medications when the antidepressants that were being used failed to get a positive change in the person. The majority of drug trials on Rexulti have been to study efficacy in treating major depressive disorder. The FDA granted approval for this use when Rexulti first appeared on the market. Notably, much research shows that only a percentage of persons with MDD adequately respond to antidepressant medications, according to a mammoth review of medical literature published in 2015 in the Journal of Psychiatry and Neuroscience.1
This body of research showed that about half of all controlled antidepressant drug trials ended up with negative results. And, that completed and attempted suicide rates actually doubled compared to the placebo group in drug trials.
Other published research has similarly shown that antidepressants are significantly associated with worsened depression and suicidality. Some researchers whose studies are funded by drug companies protest when others vilify antidepressants as too dangerous. Yet, other independent researchers claim that antidepressants should not be prescribed to anyone as their risk-to-benefit ratio is too high. Perhaps these problems inspired drug companies to look for a solution by combining an antipsychotic drug with traditional antidepressants for a better outcome.2-5
What about Rexulti Long-Term Effects in Schizophrenia Trials?
There have been numerous short-term but only a few long-term studies on Rexulti for the treatment of schizophrenia. Research published in the 2018 Journal of the Mental Health Clinician, describes 2 short-term (6-week) trials that were then extended to 52 weeks of monotherapeutic (Rexulti) schizophrenia treatment. Participants were all diagnosed with exacerbated schizophrenia, requiring hospitalization.
In the BEACON 6-week trial, 657 participants were divided into 4 groups:
- 184 placebo
- 120 were given 1 mg Rexulti daily
- 186 were given 2 mg Rexulti daily
- 184 were given 4 mg Rexulti daily
In the VECTOR 6-week trial, 623 participants were divided similarly:
- 184 placebo
- 90 were given .25 mg Rexulti daily
- 182 were given 2 mg Rexulti daily
- 180 were given 4 mg Rexulti daily
Trial results were remarkably similar. In both trials, participants reported Akathisia most frequently at 2mg and 4mg doses. Also in both of these short-term trials, weight gain doubled or tripled in all the Rexulti groups, compared to the placebo groups.
The extended 52-week study used variable dosages from 1 mg to 6 mg daily. At the end of the trial, researchers reported that only 34% of the participants completed the trial and that 58.6% of participants experienced at least one adverse event.6
The FDA drug label also describes two 6-week Rexulti schizophrenia trials reported by drug makers, with similar results to the above.
Rexulti Clinical Trials as an Adjunct to Antidepressant Medications
 The FDA drug label cites a total of two 6-week-long studies combining Rexulti with antidepressant medications in patients who did not respond well to antidepressants alone. About 600 participants were treated with combined Rexulti (at varying dosages) plus an unspecified antidepressant, compared to approximately 400 participants (the study was double-blind) who were given a placebo plus their regular antidepressant medication.7
The FDA drug label cites a total of two 6-week-long studies combining Rexulti with antidepressant medications in patients who did not respond well to antidepressants alone. About 600 participants were treated with combined Rexulti (at varying dosages) plus an unspecified antidepressant, compared to approximately 400 participants (the study was double-blind) who were given a placebo plus their regular antidepressant medication.7
Surprisingly, a searching review of the drug label lists no positive results supporting this protocol for the treatment of depression. In addition, the antidepressant trials found that there was a greater shift from “normal” to “abnormal” for extrapyramidal and akathisia symptoms in the Rexulti group, compared to the placebo group.
These results were mirrored in the other trials in the schizophrenia population.
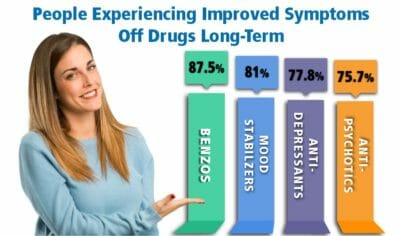
Of additional concern, the incidences of akathisia and restlessness increased when Rexulti dosages were increased. Below is a summary of the short-term and long-term effects of Rexulti that clinicians have reported in clinical trials.
 Although it is classified as an antipsychotic drug, one of the common uses of Rexulti is to prescribe it on top of antidepressant medications when the antidepressants that were being used failed to get a positive change in the person. The majority of drug trials on Rexulti have been to study efficacy in treating major depressive disorder. The FDA granted approval for this use when Rexulti first appeared on the market. Notably, much research shows that only a percentage of persons with MDD adequately respond to antidepressant medications, according to a mammoth review of medical literature published in 2015 in the Journal of Psychiatry and Neuroscience.1
Although it is classified as an antipsychotic drug, one of the common uses of Rexulti is to prescribe it on top of antidepressant medications when the antidepressants that were being used failed to get a positive change in the person. The majority of drug trials on Rexulti have been to study efficacy in treating major depressive disorder. The FDA granted approval for this use when Rexulti first appeared on the market. Notably, much research shows that only a percentage of persons with MDD adequately respond to antidepressant medications, according to a mammoth review of medical literature published in 2015 in the Journal of Psychiatry and Neuroscience.1 The FDA drug label cites a total of two 6-week-long studies combining Rexulti with antidepressant medications in patients who did not respond well to antidepressants alone. About 600 participants were treated with combined Rexulti (at varying dosages) plus an unspecified antidepressant, compared to approximately 400 participants (the study was double-blind) who were given a placebo plus their regular antidepressant medication.7
The FDA drug label cites a total of two 6-week-long studies combining Rexulti with antidepressant medications in patients who did not respond well to antidepressants alone. About 600 participants were treated with combined Rexulti (at varying dosages) plus an unspecified antidepressant, compared to approximately 400 participants (the study was double-blind) who were given a placebo plus their regular antidepressant medication.7 Increased mortality including risk of stroke, cerebral adverse events, and death in elderly dementia patients
Increased mortality including risk of stroke, cerebral adverse events, and death in elderly dementia patients We tailor a treatment program that is highly individualized to fit the needs of the client. That will usually involve
We tailor a treatment program that is highly individualized to fit the needs of the client. That will usually involve 







